New Research Reveals Pancreatic Cancer Patients Who Receive Precision Medicine Live An Average Of One Year Longer Than Those Who Do Not
Pancreatic Cancer Action Network and Perthera Study in Lancet Oncology Underscores the Urgent Need for Every Pancreatic Cancer Patient to Get Tested Through a Molecular Profiling Platform
MANHATTAN BEACH, Calif. and HOLLISTON, Mass., March 2, 2020 /PRNewswire/ — The Pancreatic Cancer Action Network (PanCAN) and Perthera, Inc., announced a pivotal paper published today in Lancet Oncology highlighting the importance of selecting treatments based on pancreatic cancer patients’ tumor mutations. This is the first study to demonstrate an overall survival benefit from precision medicine in pancreatic cancer patients.
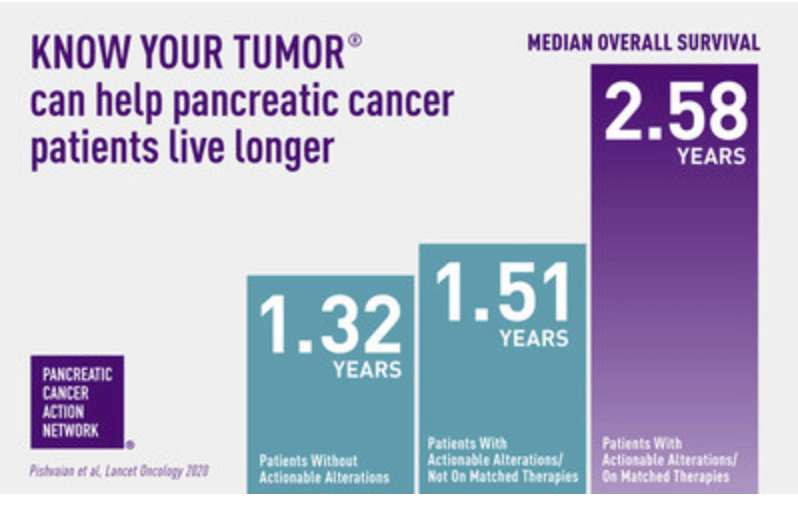
The study analyzed more than 1,000 pancreatic cancer patients enrolled in PanCAN’s Know Your Tumor® precision medicine service, powered by Perthera, and found those patients who received matched therapies following molecular profiling of their tumor, using the Perthera Platform, saw an overall survival benefit of one year longer than those who did not.
Pancreatic cancer is the world’s toughest cancer with an overall five-year survival rate of just 10 percent. It is currently the third leading cause of cancer-related death in the U.S., underscoring the urgent need for new treatment options and research discoveries in this space.
When PanCAN’s Know Your Tumor service was started with Perthera in 2014, key precision medicine resources such as molecular profiling were rarely offered to patients with pancreatic cancer because many doctors assumed there wasn’t anything actionable to be found. The study showed that approximately 25 percent of pancreatic cancer patients have an “actionable alteration.” It also underscores the significance of the most recent National Comprehensive Cancer Network (NCCN) guidelines that state that every patient with pancreatic cancer should get testing – both molecular profiling of their tumor and germline (genetic) testing for alterations they were born with.
“The results of this study will help re-write the future for cancer patients for years to come,” said Gary Gregory, CEO & President, Perthera, Inc. “Increasing patient survival is the cornerstone of the work that PanCAN has spearheaded with Perthera across the U.S. This study clearly proves that, by utilizing precisely matched therapies provided by molecular profiling, along with Therapeutic Intelligence and a Molecular Tumor Board, patients with pancreatic cancer have experienced significant increases in both overall and progression-free survival.”
“This is critical news for pancreatic cancer patients. Every tumor is different and through this study, we know that patients who get tested and receive treatment based on their tumor’s biological characteristics are living longer,” said Lynn Matrisian, PhD, MBA, Chief Science Officer at PanCAN and an author on the paper. “It is a strong reminder to healthcare professionals to offer tumor profiling to all their pancreatic cancer patients. And it will further provide an incentive to the scientific community to pursue new targeted treatments for even more pancreatic cancer patients.”
Several of the molecularly-matched therapies given have now been FDA approved for patients with pancreatic cancer based on select biomarkers. Adopting molecular profiling into routine practice will be critically important to making sure these patients do not miss out on life-extending opportunities that can now be covered by insurance. “These real-world outcomes suggest that the adoption of a precision medicine platform can have a substantial impact on survival in patients with pancreatic cancer, and that molecularly-guided treatments targeting oncogenic drivers and the DNA damage repair pathway warrant further prospective evaluation,” said Mike Pishvaian, MD, PhD, Perthera’s Chief Medical Officer GI Medical Oncology, University of Texas, MD Anderson Cancer Center, Johns Hopkins Medicine.
PanCAN recommends that all pancreatic cancer patients undergo testing of both their tumor tissue (molecular profiling) and blood or saliva for genetic (germline) changes to determine if they have an “actionable alteration” and to identify treatment options for that patient. Patients can enroll in PanCAN’s free Know Your Tumor precision medicine service today, as well as receive free, in-depth, and personalized resources and information on the disease through PanCAN’s Patient Central.
The Perthera Platform has been proven to capitalize upon “actionable alterations” and improve patient outcomes across numerous, peer reviewed clinical publications. The Perthera Platform, which has been used by over 250 cancer treatment sites across the U.S., captures a patient’s entire medical and treatment history, as well as their multi-omic molecular profile, to create a personalized treatment plan, that enable physicians to effectively harness the power of precision medicine.